is liquid bromine a homogeneous mixture
There is only one phase of matter observed in a homogeneous mixture. Sulphur and common salt.

Matter And Change Mrs Coyle Chemistry A Classification
B After separation bromine cannot be broken down into simpler substances.

. B The Assertion and the Reason are correct but the. Which of the following is a mixture. Homogeneous mixtures are also known as solutionsWhen you think of a solution you probably think of a liquid.
Indicate the physical state for each of the following elements at 25 C and normal pressure. D Bromine Br is on the right side of the periodic table. Name the mixture whose particles are large enough to scatter light.
Brass is an example of a a homogeneous compound b homogeneous mixture c. Iron filings and sulphur. B Because its composition is not uniform throughout orange juice is a heterogeneous mixture.
Bronze is an alloy of copper zinc and tin. The formula is C12H22011 LOC. A air b brass c iodised table salt d steel.
A heterogeneous mixture of. Mixture which has uniform composition for example. C Carbon tetrachloride has the properties of carbon and chlorine.
A mixture of can be separated by magnetic separation. A homogeneous liquid mixture of bromine Br2 and carbon tetrachloride CCl4 b. Silver bromide compound is made of silver and bromine elements.
Bromine is heavy and it keeps. A homogeneous mixture composed of two or more substances in which a mixture a solute is a substance dissolved in another substance known as. D Bromine and carbon tetrachloride can be separated by a separating.
So it is either a compound like hexane. It is not chemically pure. Graphite an allotrope of carbon Question.
A Aluminum oxide is a single chemically pure compound. A homogeneous mixture is a mixture in which the composition is the same throughout Soda water is a solution. But homogeneous solution is particularly used to denote solid to liquid mixture or liquid to liquid mixture.
Naphthalene and common salt. Brass is an alloy of copper and zinc. An example of a liquid metal and a liquid non-metal is a gallium mercury b mercury chlorine c mercury bromine d bromine sulphur.
Figure 45 Metals Nonmetals. 7 32 F is the same temperature as 100 C. A mixture in which the constituents are uniformly distributed throughout without any clear boundary of separation is called.
A It is a solid-liquid homogeneous mixture. Identify the statements which comply with bromine dissolved in carbon tetrachloride. B After separation bromine cannot be broken down into simpler substances.
A Homogeneous mixture b Non-Homogeneous mixture. A It is a solid-liquid homogeneous mixture. Solid Homogeneous Mixture Examples.
A clear liquid cant be an element because the only elements liquid at room temperature are bromine which is dark brown and mercury which is metallic. 10 The units of specific heat are cal gF. Classify each element as a metal nonmetal or semimetal.
It is uniform in composition throughout. It is a. As well as liquid.
A bromine atom gains that electron to become bromide ion. A barium b boron c bismuth d bromine. A homogeneous mixture can be defined as a mixture that has a uniform composition throughout its mass.
Salt solution and sugar solution are examples of homogeneous mixtures. C Carbon tetrachloride has the properties of carbon and chlorine. Homogeneous Mixtures Heterogeneous Mixtures centrifugation coagulation distillation evaporation filtration hand picking magnetic separation sieving winnowing sedimentation Mixture Separation Techniques True solution Colloidal solutions Suspensions These.
9 Carbohydrates and proteins have the same caloric value per gram. Answer 1 of 3. A clear liquid also cant be a heterogeneous mixture or it would not be clear.
A homogeneous solution of sugar dissolved in water O E. When bromine and hydrogen undergo following reaction-. Mixtures A mixture is a combination of two or more pure substances in which each pure substance retains its individual chemical properties.
We hope the above multiple choice questions for Class 9 Social Science for Chapter 2 Is Matter Around Us Pure provided above with answers based on the latest. Liquid in solid e. 8 The energy in chemical bonds is kinetic energy.
Which out of the following is a heterogeneous mixture. Ii Homogeneous mixture A mixture which has uniform composition throughout is called a homogeneous mixture. B A homogeneous solid solution of two or more metals is called an alloy.
Mixture whether they are homogeneous or heterogeneous How would you separate mixtures. Solid in solid mixture 5. Silver bromide is a pure substance.
However many solids are also considered homogenous mixturesThere is a wide variety of solid homogeneous mixtures from naturally occurring materials like stone to synthetic plastics. A Tin Sn is a homogeneous mixture of sulfur S and nitrogen N b Table salt is a homogeneous mixture of sodium metal and chlorine gas CORRECT-- cThe air trapped in a bicycle tire is a homogeneous mixture d Sugar dissolves completely in water to give a heterogeneous mixture e Water H2O is a homogeneous mixture containing hydrogen and. A homogeneous mixture is a gaseous liquid or solid mixture that has the same proportions of its components throughout a given sample.
11 Condensation occurs when a liquid is converted to a solid. Bromine a liquid with the formula Br2 O B. Solution for Given the following combinations.

Pure Substances A Sample Of Matter That Has Definite Chemical And Physical Properties Ppt Download

Chapter 2 Properties Of Matter Jennie L Borders

Teachers Notebook Matter Worksheets Science Teaching Science
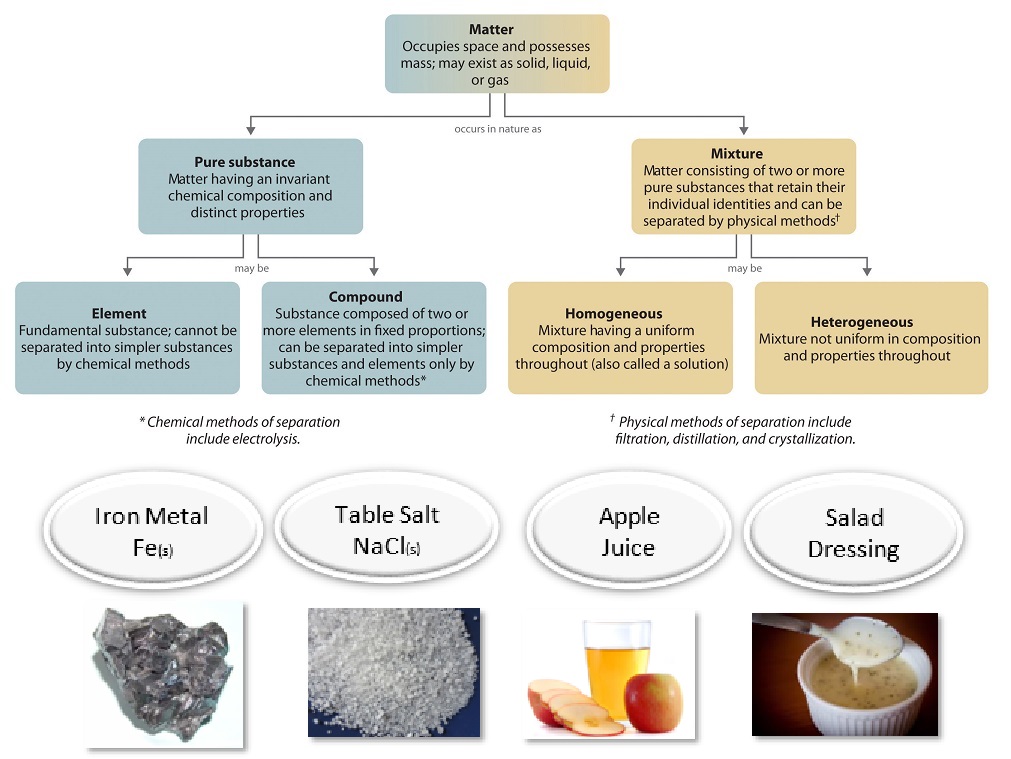
3 05 Pure Substances And Mixtures Chemistry Libretexts
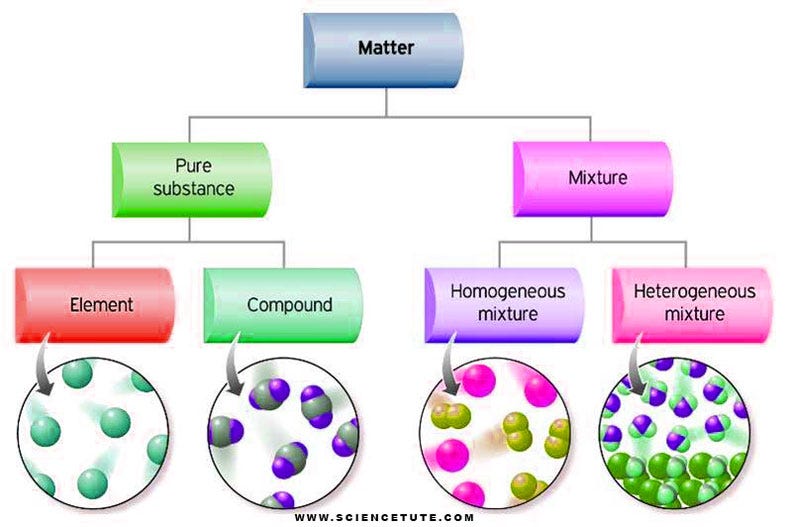
A Z Of Classification Of Matter What Is The Classification Of Matter By Science Books Medium

Classification Of Matter Matter Chemistry Is The Study

Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Matter Worksheets

Homogeneous And Heterogeneous Mixture Heterogeneous Mixture Homogeneous Mixture Mixtures

Homogeneous Mixture Homogeneous Mixture Easy Science Chemistry Classroom

Is Table Salt A Homogeneous Or Heterogeneous Mixture

Give Three Differences Between Homogeneous Mixture And Heterogeneous Mixture Brainly In

Homogeneous Heterogeneous Mixture Definition Examples Selftution

Heterogeneous And Homogeneous Mixtures What S The Difference Homogeneous Mixture Heterogeneous Mixture Examples Of Mixtures

Chapter 4 Elements Compounds And Mixtures Oh My
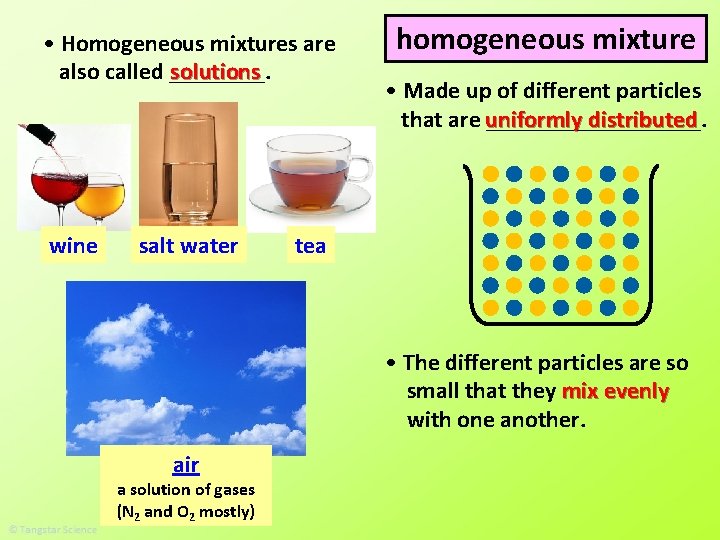
Classifying Matter How Can Matter Be Classified Matter

Iq 1 1 Define Matter 2 What 2 Major Groups Is Matter Divided Into 3 Give An Example Of A Heterogeneous And Homogeneous Mixture Ppt Download

What Is A Homogeneous Mixture Definition And Examples Homogeneous Mixture Heterogeneous Mixture Compounds And Mixtures

Pure Substances And Mixtures Compounds And Mixtures Homogeneous Mixture Heterogeneous Mixture

Homogeneous And Heterogeneous Mixtures Card Sorting Activity Heterogeneous Mixture Sorting Activities Sorting Cards
0 Response to "is liquid bromine a homogeneous mixture"
Post a Comment